Nitrosonoma bacteria – Sourced from Michigan state
Nitrosomonas
Sadly very little is mentioned of Nitrosomonas in a ideal metabolism/input:output performance BUT I did find it interesting that N. Samonas (shorthand) does require some Fe ( iron ).
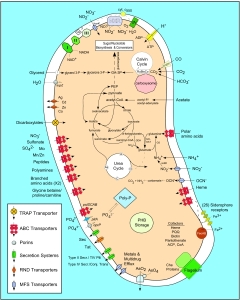
One of the more interesting and unexpected findings to come out of the genome is insight into the strategy of N. europaea to accumulate Fe from the environment. These bacteria have a large appetite for Fe given all of the cytochromes they produce. Therefore, it was not surprising to learn that receptors for Fe-siderophores are encoded. The diversity of different classes of receptors and the number of genes devoted to these receptors (>20) was something of a surprise. However, even more surprising was the almost complete absence of biosynthetic pathways for siderophores. An exception is the citrate transporter, which utilizes a product and intermediate of the TCA cycle. Apparently, N. europaea relies on other bacteria to produce siderophores and then uses an arsenal of receptors to harvest their products. This reliance on other bacteria for siderophore production is in contrast to the otherwise complete self-reliance of this bacterium, which can extract energy and produce complex cellular constituents from simple inorganic nutrients. It is likely that N. europaea seeks to manage the Fe nutrition of its environment as a survival strategy (much like some pseudomonads on plant surfaces or pathogens in hosts)
— http://www.ncbi.nlm.nih.gov/pmc/articles/PMC154410/
Unfortunately there doesn’t seem to be a quantification of HOW large “a large appetite for Fe…” is specifically. I do have it in mind to add ~10ml of MaxiCrop w/Iron chelate into the system and see what the ppm performance change is/if any.
Not exactly useful but I did find the journey to NH3 somewhat interesting
NH₃ + O₂ + 2H+ + 2e− → NH₂OH + H₂O → NO₂− + 5H+ + 4e−
I’m no chemist so I will get somethings wrong
- NH₃ (Ammonia) PLUS
- O₂ (dioxygen) PLUS
- 2H- (two elemental atoms of ionized hydrogen[1], TWO separate atoms and not a molecule ) PLUS
- 2e- (two lost/freed electrons where the – in 2e- is meant to denote a negative charge )
- Becomes (→)
- NH₂OH ( Hydroxylamine [2] )PLUS
- H₂O (water)
- FINALLY becomes (→)
- NO₂- ( Nitrite ) AND
- 5H+ (5 ionized hydrogen atoms ) AND
- 4e- with four electron’s lost in the process.
“`
Ionized hydrogen [1]
Hydroxylamine [2]
Line 10 of the chemical process is especially interesting as it shows why PH ( free hydrogen in solution ) drops as NH4/NH3 is consumed in the Nitrogen cycle by the introduction of 5 positively charged hydrogen atoms. If I’m reading this right, 5H+ isn’t literally ionized hydrogen but more likely H3O/peroxide ( http://en.wikipedia.org/wiki/Hydrogen_atom#Hydrogen_ion ).
Nitrification cycle efficiency
When
2.2 mg-wet cell/ml of N. europaea in phosphate buffer
(pH 7.5) were incubated with a continuous supply of
air or oxygen, NH4+ –N in 200 ml solution was oxi-
−dized to NO2 –N at 47 and 37% conversion efficien-
cies, respectively, in 52 h (Fig. 1).
— Department of Bioengineering, Tokyo Institute of Technology – Page 3 – Section #3 Department of Bioengineering, Tokyo Institute of Technology,
Well then… in perfect laboratory conditions, Nitrosomonas & Nitrobacter converts less than half of the available Ammonia into NO3. So worse then a 2:1 ratio and closer to 5:2